Abstract
Background: Vimentin (VIM) is a type III intermediate filament (IF) that anchors organelles in the cell, maintains cell integrity, and facilitates mitosis. A 2018 study demonstrated that the up-regulation of VIM is associated with poor clinical outcome in patients with Acute Myeloid Leukemia (AML). This study also reported that VIM regulates signaling networks that promote leukemic cell survival while conferring resistance to various stressors. A separate study revealed that VIM over-expression was associated both with markedly shorter overall survival (OS) and leukemia-free survival (LFS) in adult AML patients. VIM has been shown to be targetable by the steroidal lactone Withaferin-A which directly binds VIM, hyper-phosphorylates VIM's Serine 38 residue, and leads to an anti-proliferative effect characterized by perturbation of intermediary filaments. We investigated VIM expression and prognostic impact in pediatric AML, and whether inhibiting VIM affected chemosensitivity in vitro.
Methods: Reverse Phase Protein Array (RPPA) was performed to quantify relative expression of 233 total and 63 post-translational modified proteins in 500 pediatric AML patients from the COG AAML1031 trial. AML cell lines with low VIM expression (OCI-2) and high VIM expression (OCI-3) as well as fresh, primary AML samples were exposed to the VIM inhibitor Withaferin-A alone, or in combination with Doxorubicin and Cytarabine chemotherapy. Other known VIM inhibitors including FiVe1, Simvastatin, and Ganoderic Acid A were tested for efficacy in our context, but only Withaferin-A produced a promising anti-leukemia effect.
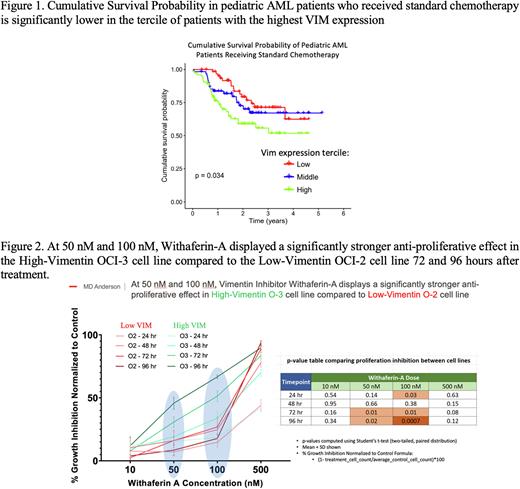
Results: In pediatric AML, VIM was over-expressed relative to normal CD34+ cells in all subtypes: bulk, CD3/CD19 depleted, CD34+, CD34+/38+, CD34+/38-, CD34-, CD34-/38+, and CD34-/38-. Pearson correlation analysis revealed significant negative correlations between VIM and proteins known to decrease during metastasis such as PDGFR-beta, TP15.pS15, HIF1-alpha and VHL. Elevated VIM correlated positively with HNRNPK, WTAP, ASH2L, and KDM1A, and H3K4Me2; alterations in histone methylation that are associated with metastatic potency. VIM expression was highest in FAB subtypes M4 and M5, and in patients with t(9;11) and MLL-rearrangements. VIM levels were lower in cytogenetically favorable and higher in unfavorable risk groups. Cumulative survival probability to standard chemotherapy progressively decreased as VIM expression increased (Fig. 1). We tested whether VIM inhibition with Withaferin-A at 50 nM and 100 nM, would affect proliferation or cell survival, finding a significantly stronger anti-proliferative effect (p<0.02) in the high-VIM OCI-3 cell line compared to the low-VIM OCI-2 cell line at 72 and 96 hours after treatment (Fig. 2). Combined with chemotherapy, Withaferin-A lead to greater cell death in the high-VIM OCI-3 cell line as opposed to low-VIM OCI-2 at 72 and 96 hours. For instance, in OCI-3, 100 nM Withaferin-A combined separately with either 1 uM Cytarabine or 50 nM Doxorubicin to decrease proliferation more than either chemotherapeutic agent alone (p=0.007 and p=0.0013). In primary AML samples, proliferation was slightly more inhibited at 72 hours post-treatment when Withaferin-A was paired with individual chemotherapeutics.
Conclusions: Proteomic analysis demonstrated that VIM was elevated in numerous leukemia cell subtypes relative to normal CD34+ cells in children and adults and that prognosis worsened as VIM levels increased in pediatric AML. AML cell lines highly expressing VIM such as OCI-3 displayed sensitivity to both Withaferin-A monotherapy and Withaferin-A in combination with Cytarabine and Doxorubicin. Finally, Withaferin-A significantly reduced viability and proliferation after 72 hours of treatment in primary AML samples at select doses in combination with chemotherapeutics. These results suggest that targeting VIM could improve the efficacy of chemotherapy in high VIM expressing subpopulations and potentially improve overall survival. Withaferin-A's efficacy both as a mono-therapeutic and in combination with standard chemotherapeutics used to treat AML patients offers an exciting new area of research that requires further investigation.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal